The C-C and C-H bonds are nonpolar and the least reactive in traditional ionic reactions. We will discuss this later, but as far as the carbon-halogen bonds and dipole moments, keep in mind that these are all considered polar covalent bonds. This is because of polarizability which is the characteristic of how the electron cloud around an atom responds to changes in the electronic environment. However, the C-I bond is very reactive, and we put partial charges on the bonded atoms. Now, while the C-F will by all means be considered a polar bond (1.5-unit difference), the C-I bond does not fit in this definition (0.2-unit difference). Anything of less difference is considered a nonpolar bond.įor example, alkyl halides are the compounds you will see a lot in organic chemistry, particularly in substitution and elimination reactions. This often causes confusion however, it is accepted that a bond is polar when the electronegativity difference between two atoms is ≥ 0.5 units. So, you may wonder what “large” and “little” difference is, and how large the electronegativity difference should actually be to consider a bond polar or ionic. Here is a table with the electronegativity values of the most commonly encountered elements in organic chemistry: Remember the trends of electronegativity it increases across a row of the periodic table ( left to right), and going up a column of the periodic table. Now, the ionic bonding occurs when the difference in electronegativities is very large and one of the atoms completely takes the bonding electrons. Polar covalent bonds and dipole moment originate when there is a significant difference in the electronegativities of the two bonded atoms. 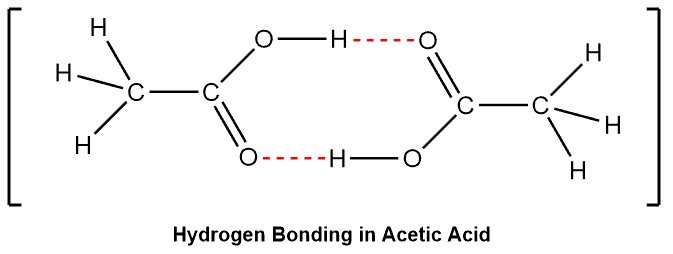
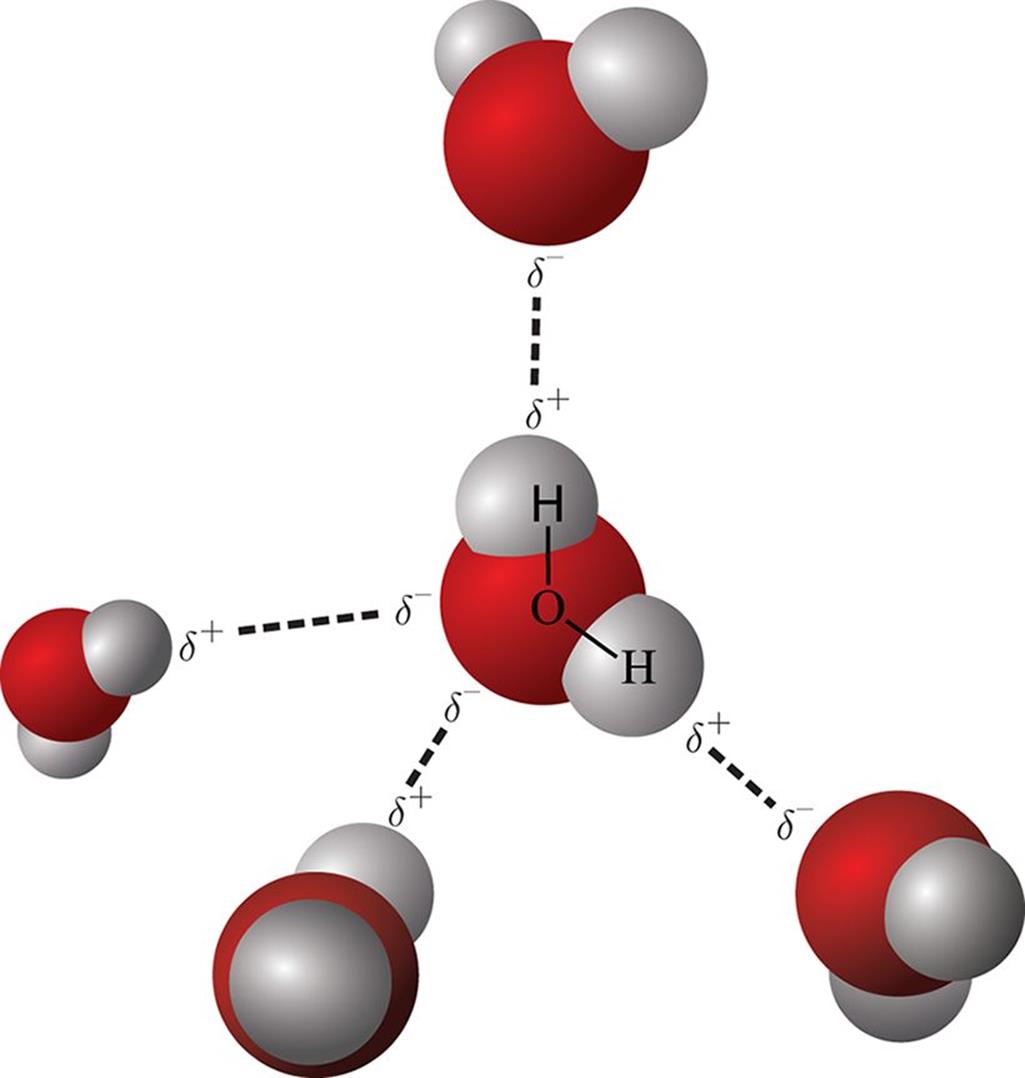
How do I know if it is Ionic or Polar Covalent? The arrow starts from the less electronegative element and points toward the more electronegative element as shown above. The direction and magnitude of polarity in a bond are given by the dipole moment which is indicated by an arrow. Mainly, the more electronegative atom bears a partial negative, and the other atom has a partial positive charge which are indicated by the delta plus (δ+) and delta minus (δ-) symbols: There is a higher density towards the more electronegative element and as a result, both elements have partial charges. To understand the nature of dipole-dipole interactions, remember that when two atoms with different electronegativities are connected, we have a polar covalent bond, and the shared electron pair of the covalent bond is not in the middle of the two atoms. Now, most organic compounds are not ionic, and therefore, we will focus more on the intermolecular interactions between the covalent compounds. We will discuss the relationship between intermolecular forces and physical properties later, but for now, keep in mind that the stronger these interactions the higher the melting and boiling points are. In other words, there is not really such thing as one bond between two ions/atoms like in covalent compounds and that’s why they need a lot of thermal energy to break these interactions and therefore, have high melting points. Unlike the covalent bonding, ionic bonds are not directional and specific between given atoms, but rather they form a lattice as an interaction among the entire cluster of the ions: 
What is important here is that we are not talking about the interaction of one Na + and Cl + ion together. 
Among other interesting features and applications, we learn that it has a melting point of 801 ☌ which is extremely higher than the typical range for organic (covalent) compounds. For example, sodium chloride (NaCl) is one of the first ionic compounds we talk about in general chemistry. All of them are electrostatic interactions meaning that they all occur as a result of the attraction between opposite charges and which of these forces is present or predominates in a given compound, depends on its functional groups.īack from general chemistry, we know from the structure of salts that oppositely charged particles tend to show very strong electrostatic interactions and these are the ionic compounds. Dipole-dipole, London dispersion (also known as Van der Waals) interactions, hydrogen bonding, and ionic bonds are the main types of intermolecular interactions responsible for the physical properties of compounds.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
